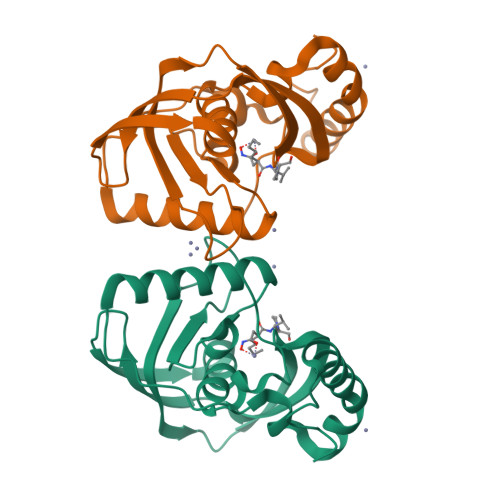
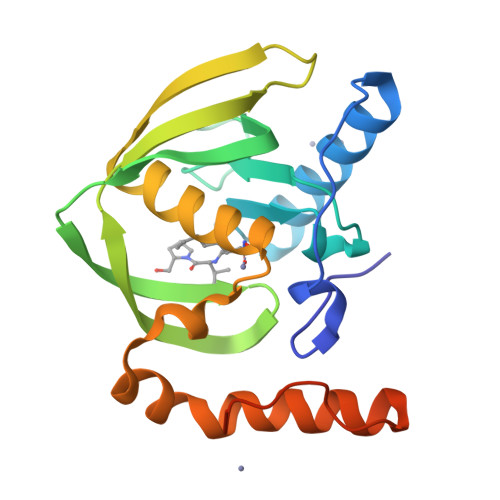
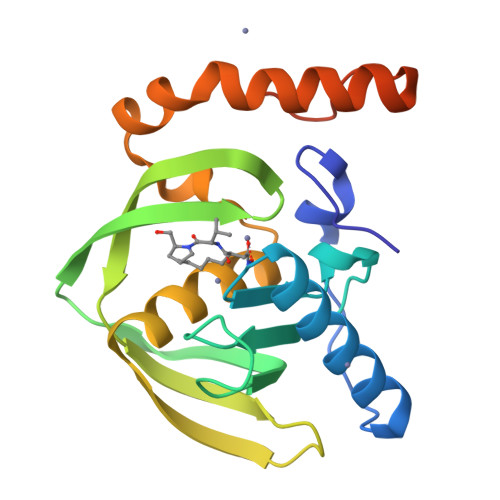
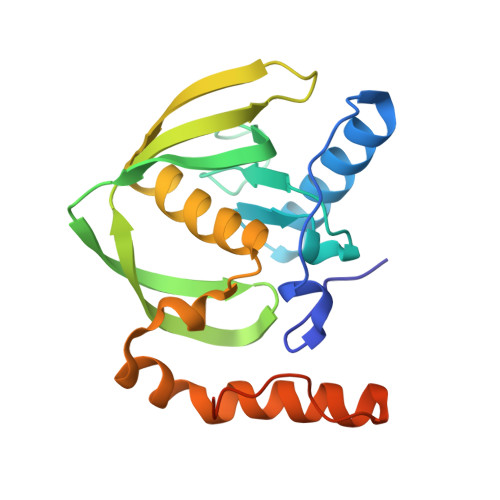
Trapping conformational states along ligand-binding dynamics of peptide deformylase: the impact of induced fit on enzyme catalysis
Fieulaine, S., Boularot, A., Artaud, I., Desmadril, M., Dardel, F., Meinnel, T., Giglione, C.(2011) PLoS Biol 9: e1001066-e1001066
- PubMed: 21629676
- DOI: https://doi.org/10.1371/journal.pbio.1001066
- Primary Citation of Related Structures:
3M6O, 3M6P, 3M6Q, 3M6R, 3O3J, 3PN2, 3PN3, 3PN4, 3PN5, 3PN6 - PubMed Abstract:
For several decades, molecular recognition has been considered one of the most fundamental processes in biochemistry. For enzymes, substrate binding is often coupled to conformational changes that alter the local environment of the active site to align the reactive groups for efficient catalysis and to reach the transition state. Adaptive substrate recognition is a well-known concept; however, it has been poorly characterized at a structural level because of its dynamic nature. Here, we provide a detailed mechanism for an induced-fit process at atomic resolution. We take advantage of a slow, tight binding inhibitor-enzyme system, actinonin-peptide deformylase. Crystal structures of the initial open state and final closed state were solved, as well as those of several intermediate mimics captured during the process. Ligand-induced reshaping of a hydrophobic pocket drives closure of the active site, which is finally "zipped up" by additional binding interactions. Together with biochemical analyses, these data allow a coherent reconstruction of the sequence of events leading from the encounter complex to the key-lock binding state of the enzyme. A "movie" that reconstructs this entire process can be further extrapolated to catalysis.
Organizational Affiliation:
CNRS, ISV, UPR2355, Gif-sur-Yvette, France.